As published by US Medicine, the Voice of Federal Medicine
by Mary Anne Dunkin | Aug 21, 2025
FORT SAM HOUSTON, TX — Lung cancer is the leading cause of cancer deaths in the United States. Early detection is vital, especially for veterans, who face a disproportionately high risk of lung cancer due to higher rates of smoking and other exposures.
About 1.4 million veterans meet current lung cancer screening criteria, making them three times more likely than the general population to qualify for low-dose computed tomography (LDCT) scans.1
While LDCT screening reduces lung cancer mortality by 16%, it frequently detects indeterminate pulmonary nodules (IPNs)—small lung masses between 6 and 29 millimeters whose nature can be uncertain. These nodules are discovered in roughly 1.6 million Americans each year and often create diagnostic uncertainty, especially in veterans, who may have nodules too small or ambiguous for biopsy.
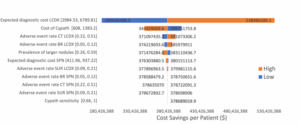
A recent study published in the Journal of Health Economics and Outcomes Research evaluated CyPath Lung, a novel diagnostic test designed to improve the evaluation of IPNs. The study’s findings highlight how this test can lead to better detection, fewer unnecessary follow-ups and substantial cost savings—an advancement with particular value for VA providers and the veterans they care for.2
Why Indeterminate Pulmonary Nodules Pose a Challenge
Indeterminate pulmonary nodules require careful management based on size and risk. The 2021 CHEST Guidelines recommend follow-up LDCT scans for nodules 6 to 8mm, regardless of risk. Nodules larger than 8 mm require more precise assessment: Low to moderate-risk nodules are typically followed with functional imaging such as PET scans. High-risk nodules (greater than 65% chance of cancer) may undergo surgical biopsy.
“Clinical assessment of such nodules is associated with relatively high uncertainty, as rates of malignancy are lower in this group, but the benefits of earlier diagnosis are substantial,” wrote Michael J. Morris, MD, of the Pulmonary/Critical Care Service at Brooke Army Medical Center and his colleagues. Veterans often fall into this diagnostic gray zone, creating a gap in care.
False positives from LDCT screening can lead to unnecessary invasive procedures, which carry risks like pneumothorax (collapsed lung) and hemorrhage. One cited study reported a complication rate of 23.8%, with associated costs ranging from $6,320 to $56,845, depending on severity. This burden disproportionately affects veterans, who already face complex health needs.
How CyPath Lung Works and Why It Matters
CyPath Lung is a noninvasive diagnostic test that analyzes sputum samples collected at home over three days. Using advanced flow cytometry and machine learning, it detects markers indicative of lung cancer.
Compared to LDCT’s specificity of about 73%, CyPath Lung demonstrated a sensitivity of 82% and specificity of 88%. In patients with small nodules (20 mm or less), sensitivity rose to 92% and specificity to 87%, with a negative predictive value of 99%, according to available data. This means the test can confidently rule out cancer in many patients, reducing the need for invasive procedures.
The study modeled one-year costs, including procedures, complications and diagnostics, comparing care with and without CyPath Lung. Results showed significant savings: Private payers could save nearly $895 million per cohort, or $6,460 per patient. Medicare savings were estimated at $379 million per cohort, or $2,733 per patient.
The authors emphasized that these savings are “primarily due to reductions in follow-up diagnostic assessments and procedures,” reducing unnecessary interventions and their risks.
They also acknowledged limitations, noting that “economic models have some limitations, most of which stem from the accuracy of the input parameters used in the model.” For example, private payer costs were estimated using Medicare multipliers, which can vary.
Procedure probabilities were based on a study of data sources only from the state of Louisiana, the authors noted. “Insofar as healthcare utilization varies by state, it is possible that [study] may not be broadly generalizable to other populations,” they wrote. “Similarly, while the results are likely to be broadly generalizable to a national population, they may not be generalizable to specific subgroups based on sociodemographic characteristics.”
Despite these caveats, they affirm that “reducing false positives and unnecessary interventions has meaningful economic and clinical value.”
Importantly, CyPath Lung is now listed on the Federal Supply Schedule (FSS/GSA Contract #36F79725D0005), making it immediately available to VA health systems nationwide. This facilitates rapid adoption across VA hospitals and clinics, ensuring veterans have access to this advanced diagnostic tool.
The study authors concluded that “integrating companion tests to work in conjunction with the standard of care has the potential to improve outcomes by more efficiently triaging patients along the care pathway, resulting in possible reductions in delay of diagnosis, more favorable prognosis, and better patient outcomes.”
- Lewis JA, Stranick A, Pennings J, Samuels LR, et al. National Survey of Lung Cancer Screening Eligibility in United States Veterans. Am J Prev Med. 2025 May;68(5):886-895. doi: 10.1016/j.amepre.2025.01.015. Epub 2025 Jan 27. PMID: 39880059; PMCID: PMC12033076.
- Morris MJ, Habib SA, Do Valle ML, Schneider JE. Economic Evaluation of a Novel Lung Cancer Diagnostic in a Population of Patients with a Positive Low-Dose Computed Tomography Result. J Health Econ Outcomes Res. 2024 Sep 17;11(2):74-79. doi: 10.36469/001c.121512. PMID: 39810799; PMCID: PMC11731590.